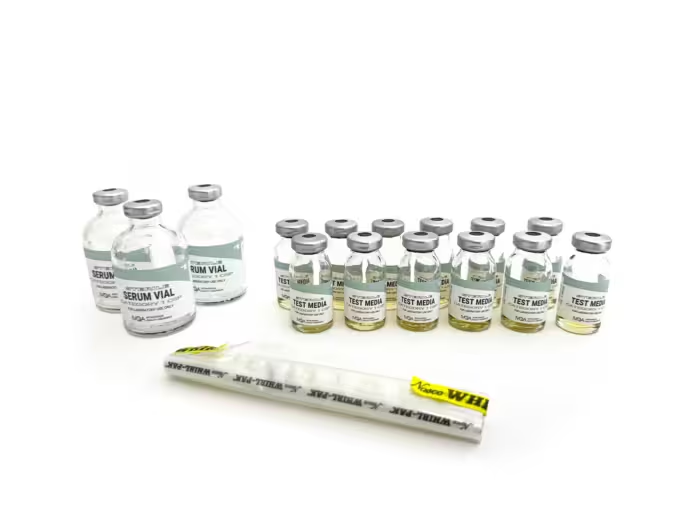
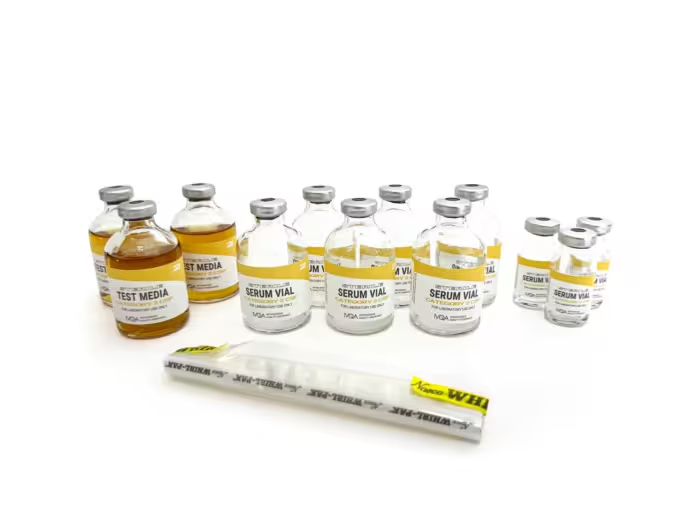
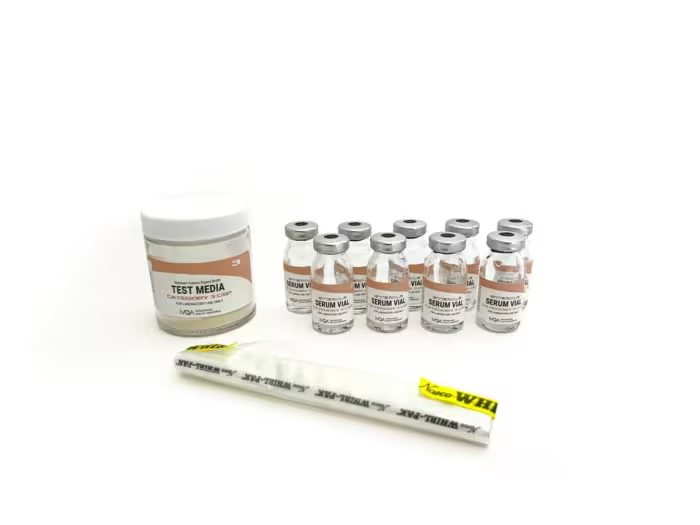
At Intravenous Quality Assurance (IVQA), we’ve been manufacturing media-fill testing supplies for over 20 years. Our mission is to make USP <797> aseptic competency straightforward, comprehensive, and repeatable. Transfer-dense workflows with triplicate endpoints deliver clear pass/fail results, while detailed, step-by-step instructions, log sheets, and a Certificate of Analysis in every kit keep teams audit-ready across Category 1, 2, and 3 CSPs.
Comprehensive, reliable, repeatable media-fill testing that ensures your team stays inspection-ready.
Finding the right kit for your application

Category 1 CSP
Category 1 CSPs involve basic and rapid compounding activities with minimal contamination risk, including single-dose vial transfers, straightforward reconstitution of powdered medications, ampule opening and contents transfer, simple IV admixtures, syringe preparation for immediate use, and sterile-to-sterile product mixing in cases where procedures are direct and expedient.

Category 2 CSP
Category 2 CSPs involve intermediate-level compounding tasks such as TPN compounding with precise multi-component mixing, multiple-step aseptic transfers, chemotherapy preparation with strict dosing, medication compounding for infusion pumps, batch preparations for several patients, and long-duration compounding processes, each demanding meticulous aseptic techniques to minimize contamination risks.

Category 3 CSP
Category 3 CSPs require advanced aseptic techniques due to their complexity and higher contamination risks, including the use of non-sterile ingredients and exposure to suboptimal air quality. Examples include bulk compounding for extended storage, incorporating non-sterile components for customized medication formulations, and utilizing sophisticated technologies for sensitive medications, all requiring rigorous sterilization measures post-compounding.