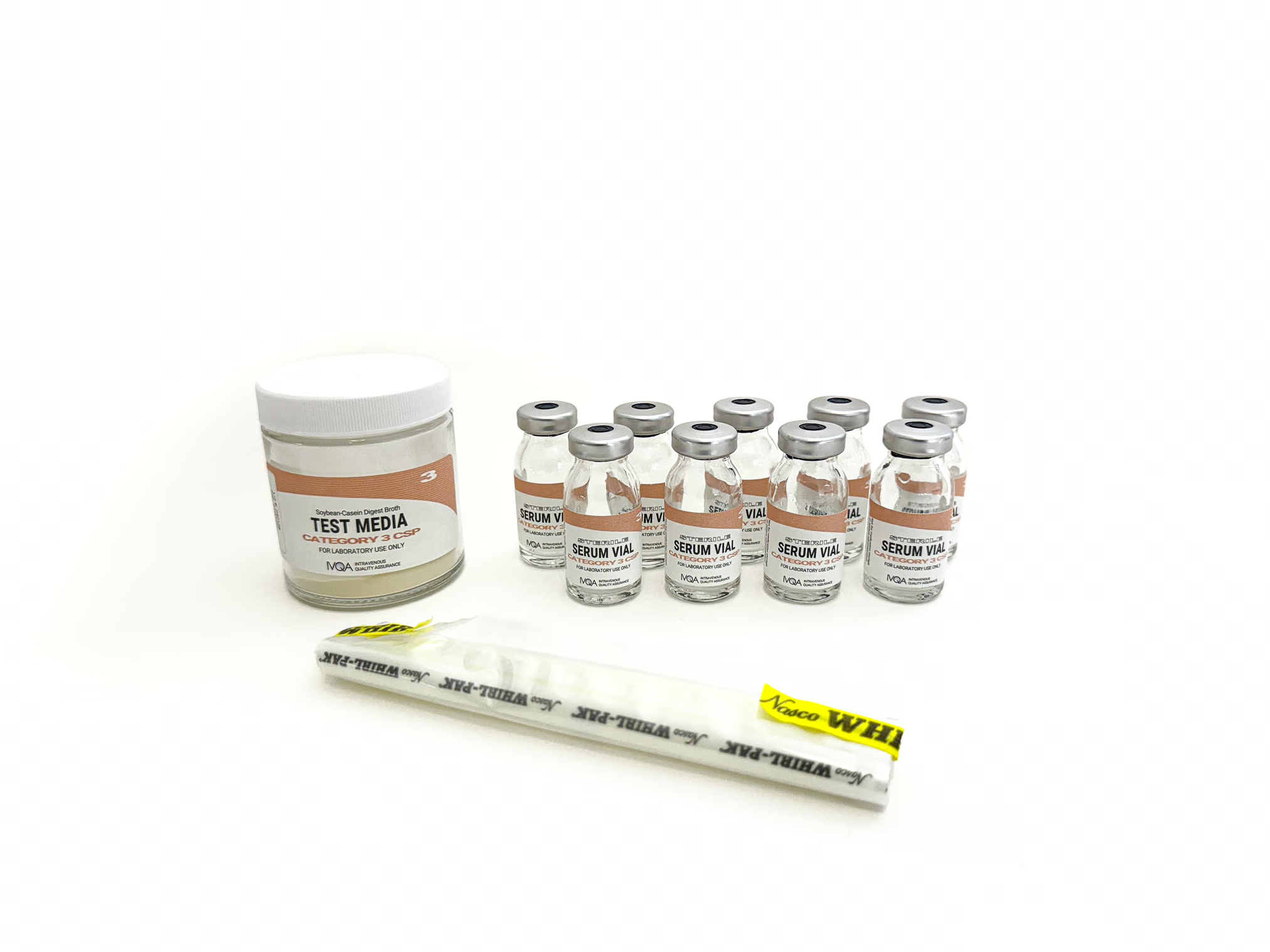
● Catalog No. MFT03
● Compliance with USP Chapter <797>
● 3-Year Expiration
● Kit Contains:
◦ Sterile 10ml Empty Serum Vials – 9 vials
◦ Tryptic Soy Broth Non-Sterile Powder (3g) – 1 container
◦ Whirl-Pak® Bag
◦ Instructions for Use, Log Sheet, & Certificate of Analysis
Category 3 CSP Media-Fill Test Kit
$74.00
USP <797> Category 3 aseptic competency—comprehensive on core manipulations and built for repeatability. Technicians reconstitute non-sterile media, perform defined transfers with 0.2 μm filtration, and complete three sets, each set producing two result vials for incubation. The higher-rigor workflow raises measured transfer count and process length, mirroring USP <797> focus on number of transfers and process complexity—providing stronger evidence of technique consistency in tighter-control programs.
IVQA media-fill test kits pair a comprehensive, transfer-dense design with illustrated, easy-to-follow Instructions for Use and an audit-ready log sheet—simplifying training and documentation. Every kit includes a Certificate of Analysis, and each lot is tested for growth promotion and sterility for dependable performance, cycle after cycle. Components arrive organized in a cleanroom-friendly thermoformed clamshell, ready to use on arrival. Use one kit per technician for onboarding and every 6 months (Category 1 & 2) or every 3 months (Category 3) thereafter. Trusted by compounding and hospital pharmacies, health-system cleanrooms, home-infusion providers, oncology/infusion centers, outsourcing facilities, and academic medical centers.
Test Procedure:
Outside an ISO Class 5 air quality environment, dissolve the 3 grams of dehydrated Tryptic Soy Broth test media by adding 100ml of non-bacteriostatic water. Replace the lid and shake vigorously to ensure the powder has completely dissolved before proceeding.

Transport the prepared non-sterile test media to an ISO Class 5 air quality environment.
Within an ISO Class 5 air quality environment, arrange the vials into three sets, each containing three empty 10ml serum vials. Label one vial from each set as ‘Control [set number]’.

Using three 30ml sterile syringes, withdraw 25ml of test media into each. Transfer 5ml of test media from set one’s syringe into 10ml vial labeled ‘Control 1’. Repeat this process for the other sets using their respective syringes, resulting in 5ml in each control vial and 20ml remaining in each syringe.
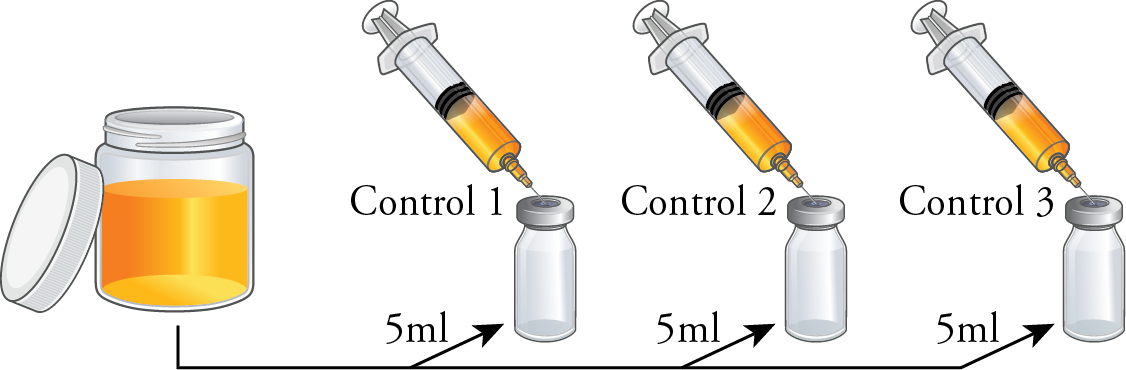
Using aseptic techniques, affix a sterile 0.2 micron porosity filter and a 20-gauge needle to each syringe. Inject 10ml from set one’s syringe into both empty 10ml vials in set one then set aside. Repeat this process for the other sets using their respective syringes and affixed filters.

Write your name and date of preparation on all nine 10ml vial labels. Then, aseptically apply a sterile adhesive seal to the rubber closures and place the sealed vials inside the provided Whirl-Pak® bag for transfer to the incubator.
Incubate the nine 10ml vials at 20°–25°C (68°– 77°F) and 30°–35°C (86°– 95°F) for a minimum of 7 days at each temperature range to detect a broad spectrum of microorganisms. A total incubation period of at least 14 days is recommended to confirm the absence of microbial growth. For the vials not labeled as ‘Control [set number]’, any signs of microbial growth such as turbidity or precipitation, at any point during the incubation period, indicates a failure in the sterility test.
Once the test is initiated:
- Must be carried out to completion without interruptions.
- Performance conditions should mimic the usual work environment.
USP Chapter <797> Category 3 CSP Guidelines
Description:
-
- Verifies aseptic techniques for Category 3 CSPs with extended Beyond-Use Dates (BUDs), adhering to the strictest USP <797> guidelines.
- Designed for high-complexity compounding, including use of non-sterile ingredients and advanced sterilization processes.
- Essential for confirming advanced aseptic competencies and environmental controls in the preparation of high-risk medications.
- Ensures advanced aseptic competencies and environmental controls are applied, especially when handling non-sterile components or exposure to environments below ISO Class 5 air quality, to achieve the highest sterility assurance levels despite the inherent complexities.
Examples:
-
-
- Preparing sterile products by incorporating non-sterile raw materials, which requires additional steps to ensure final sterility.
- Producing large volumes or quantities of compounded sterile preparations, often for future use, not limited to immediate patient-specific needs.
- Procedures that involve multiple complex aseptic manipulations or long-duration compounding, increasing the risk of microbial contamination.
- Processes where the compounding is exposed to air quality that is inferior to ISO Class 5 for an extended period, necessitating more stringent sterilization processes afterward.
- Incorporating advanced compounding technologies such as lyophilization or complex emulsification processes.
- Compounding medications that are particularly susceptible to contamination, like ophthalmic preparations or biologic therapies, especially when starting from non-sterile ingredients.
- Formulating customized medication dosages or formulations that are not commercially available, often starting from non-sterile powders or substances.
- Preparations that require terminal sterilization (e.g., filtration, autoclaving) after compounding to ensure sterility before dispensing.
-
Initial Training and Competency:
-
- Before beginning independent compounding or having direct oversight of compounding personnel, individuals must successfully complete an initial aseptic manipulation competency evaluation.
- This initial evaluation includes visual observation, media-fill testing, gloved fingertip and thumb sampling on both hands, and surface sampling of the direct compounding area.
Frequency of Ongoing Competency Testing:
-
- Aseptic manipulation competency evaluations must occur at least every 3 months for personnel compounding Category 3 CSPs.
References:
-
- USP Chapter <797> guidelines can be found in the current version of the United States Pharmacopeia and National Formulary (USP–NF), which outlines the standards for sterile compounding procedures and quality assurance practices.
- Whirl-Pak® is a registered trademark of Nasco International, Inc.
| Weight | 1.0 lbs |
|---|---|
| Dimensions | 8.8 × 8.8 × 3.8 in |